ECG can be deployed for the widespread screening of
aortic stenosis
Earlier detection and treatment will lead to better outcomes and lower costs
Underdiagnosis of aortic stenosis is a major problem
A new approach is needed to improve detection
High mortality for aortic stenosis
As high as 50% in one year1Delayed treatment increases costs
$10k additional cost per patient per year2High rate of aortic stenosis underdiagnosis
Up to 50% of AS cases are missed until post-mortem3There is a new approach to addressing underdiagnosis
AccurKardia proposes to improve timely diagnosis through systematic ECG-based screening
Unique benefits of AccurKardia's
ECG-based screening approach
Rapidly process massive amounts of preexisting ECGs
Concentrated deployment at the EHR system level
No disruption to point of care workflow
No new devices / equipment
Facilitate clinical trial enrollment for moderate and asymptomatic AS
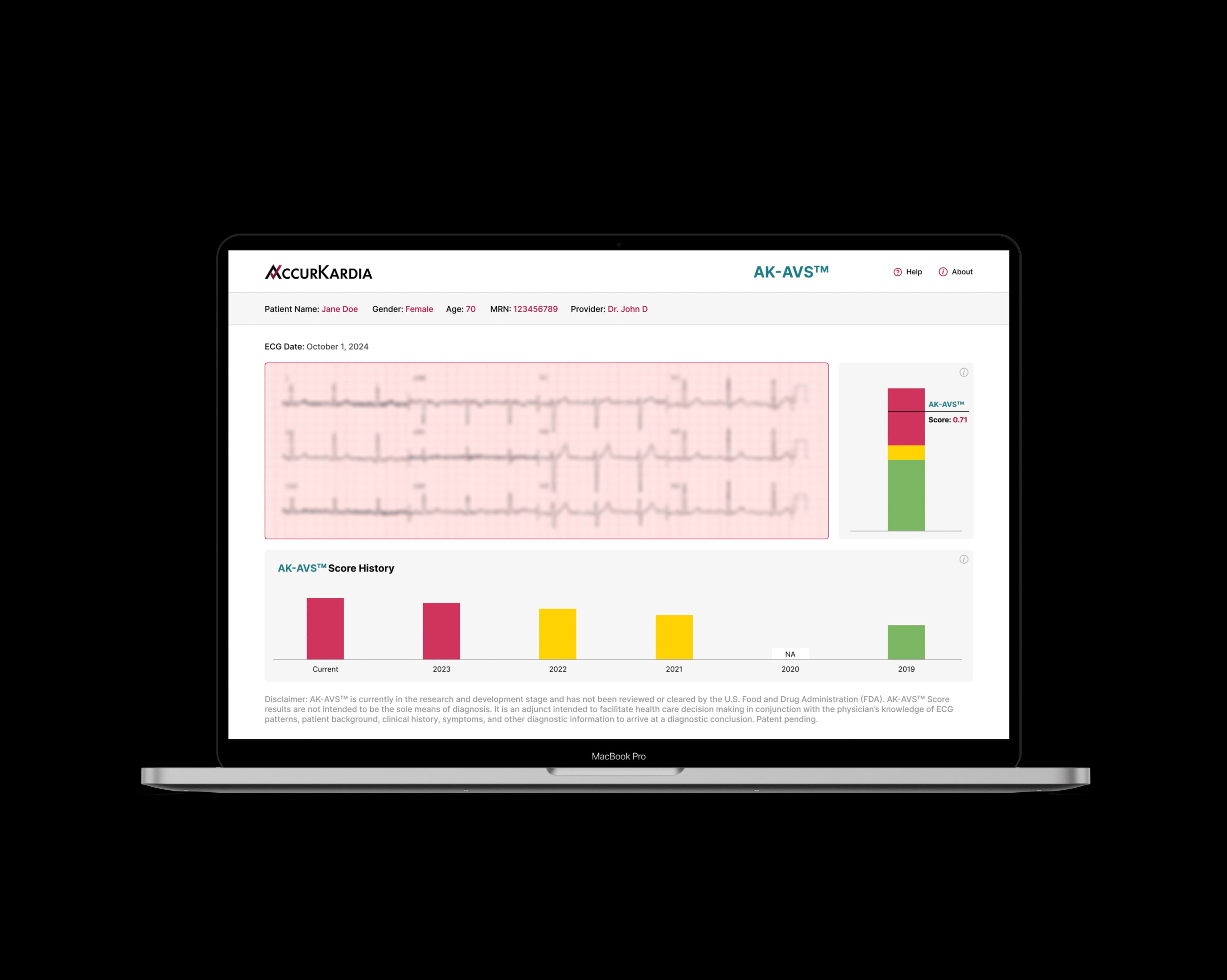
AK-AVS Performance*
74%
Sensitivity
86%
Specificity
83%
Accuracy
Download our White Paper: AK-AVS - A New Paradigm in Aortic Stenosis Detection
Gain in-depth insights into how AccurKardia’s AI-powered AK-AVS technology enhances early detection of aortic stenosis through ECG screening.
*Detection of moderate and severe aortic stenosis clinical validation based on data from Mayo Clinic Platform_Accelerate Cohort 3 Dataset from Mayo Clinic.
Remote Monitoring for Hyperkalemia:
Extending the Lab to the Home
AK+ Guard™ can process Lead I ECG data from wearables to remotely monitor hyperkalemia in at-risk patients.
AK+ Guard Hyperkalemia Model Performance*
81.2%
Sensitivity
83.2%
Specificity
89.0%
Accuracy
*Detection of moderate to severe hyperkalemia episodes based clinical validation on data from Mayo Clinic Platform_Accelerate Cohort 3 Dataset, from Mayo Clinic.
ECG can be a powerful biomarker for cardiology and beyond
AccurKardia’s ECG-based tests compare favorably to widely used diagnostics
AK-AVS = Moderate-to-Severe Aortic Valve Stenosis Detection Model
AK+ Guard = Moderate-to-Severe Hyperkalemia Detection Model
Future Pipeline
Electrolyte Dysfunctions
Amyloidosis
Heart Failure
- Bevan, G. (2019). Mortality Due to Aortic Stenosis in the United States, 2008-2017. JAMA, 2236-2238.
- Sethi, A, MD (2024). The Cost of Waiting for a Transcatheter Aortic Valve Replacement in Medicare Beneficiaries With Severe Aortic Stenosis. Structural Heart.
- Das, P. (2000). The patient with a systolic murmur: severe aortic stenosis may be missed during cardiovascular examination. QJM, 685–688.
- Collins, A. J., Pitt, B., Reaven, N., Funk, S., McGaughey, K., Wilson, D., & Bushinsky, D. A. (2017).Association of serum potassium with all-cause mortality in patients with and without heart failure, chronic kidney disease, and/or diabetes. American Journal of Nephrology, 46(3), 213–221.
- Karaboyas, A., Robinson, B., James, G., Hedman, K., Quinn, C. P. M., De Sequera, P., ... & Pecoits-Filho, R. (2021).Hyperkalemia excursions are associated with an increased risk of mortality and hospitalizations in hemodialysis patients. Clinical Kidney Journal, 14(7), 1760–1769.
- Agiro, A., Duling, I., Eudicone, J., Davis, J., Brahmbhatt, Y. G., & Cooper, K. (2022).The prevalence of predialysis hyperkalemia and associated characteristics among hemodialysis patients: The RE-UTILIZE study. Hemodialysis International, 26(3), 285–458.
- Lee, R. (2006). A meta-analysis of the performance characteristics of the free prostate-specific antigen test. Urology, 762-768.
- Lee, T. (2006). A1C Testing in the Diagnosis of Diabetes Mellitus. American Family Physician, 143-144.
- Noubiap, J. (2020). A meta‐analytic evaluation of the diagnostic accuracy of the electrocardiographic Peguero‐Lo Presti criterion for left ventricular hypertrophy. Journal of Clinical Hypertension, 1145-1153.
- Neves, P. (2017). Coronary artery calcium score: current status. Radiologica Brasileira. 182-189.
- Johns, P. (2014). Diagnosis and early detection of COPD using spirometry. Journals of Thoracic Disease. 1557-1569.
- Chou, R. (2022). Screening for Glaucoma in Adults, JAMA Network. 1998-2012
The AK-AVS™ and AK+ Guard™ are registered trademarks of AccurKardia, Inc., with all intellectual property rights owned by AccurKardia, Inc.
AK-AVS™ and AK+ Guard™ are currently for research use only and have not been cleared or approved by the US FDA for use in the United States. This technology is under development and intended solely for investigational purposes. It should not be used for clinical diagnosis, treatment, or disease screening at this time. AK-AVS™ and AK+ Guard™ are currently patent pending.
The information provided on this website is for informational purposes only and does not constitute medical, legal, or professional advice. Always consult a qualified healthcare provider for clinical decision-making.